 
In a matter of minutes, hydrogen cyanide poisoning can be fatal. It prevents nearly all of the body’s organs from using oxygen normally. Hydrogen cyanide, a chemical asphyxiant, is a systemic asphyxiant. Exothermic oligomerization occurs when distilled HCN is heated, hence it should be maintained at temperatures considerably below its melting point. Hydrogen cyanide is extremely toxic and combustible. On a large scale, HCN is used to produce potassium cyanide and adiponitrile, which are utilized in the manufacture of plastics and mining. HCN is a versatile substance since it is utilized as a highly valued precursor to a variety of chemical compounds ranging from polymers to medicines, as well as in other industrial sectors. At 25.6 ☌, it boils slightly above room temperature. The B–B bond length is 1.76 Å.HCN, or hydrogen cyanide, is a colorless, highly toxic, and combustible chemical compound. In the tenth B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. In the ninth B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. In the eighth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. There is one shorter (1.76 Å) and one longer (1.77 Å) B–B bond length. In the seventh B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. There is one shorter (1.75 Å) and one longer (1.79 Å) B–B bond length. In the sixth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the fifth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the fourth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the third B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the second B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. 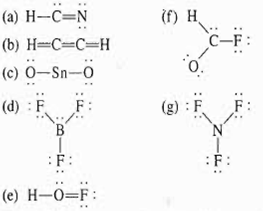
In the first B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. There are a spread of Mg–B bond distances ranging from 2.48–2.52 Å. In the fifth Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.47–2.51 Å. In the fourth Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.48–2.51 Å. In the third Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.49–2.51 Å. In the second Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share edges with three equivalent LiB12 cuboctahedra, edges with nine MgB12 cuboctahedra, and faces with eight MgB12 cuboctahedra. There are a spread of more » Mg–B bond distances ranging from 2.48–2.50 Å. In the first Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share edges with twelve MgB12 cuboctahedra, faces with two equivalent LiB12 cuboctahedra, and faces with six MgB12 cuboctahedra. There are a spread of Li–B bond distances ranging from 2.48–2.50 Å. Li is bonded to twelve B atoms to form LiB12 cuboctahedra that share edges with twelve MgB12 cuboctahedra and faces with eight MgB12 cuboctahedra. LiMg9B20 is hexagonal omega structure-derived structured and crystallizes in the triclinic P-1 space group. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
